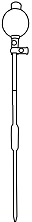Determination of Break-Point Chloride Demand
Objective:
To determine Break-Point chlorine demand in water sample.
Apparatus used:
BOD bottles, Pipette, Conical flask, Burette, Dropper, BOD incubator etc.
Solutions used Standard chlorine solution, Concentrated sulphuric acid, Potassium iodide crystals, 0.0282N Iodine solution, Starch indicator Description Chlorine Demand is the difference between the amount of chlorine added to water or wastewater and the amount of residual chlorine remaining after a given contact time.




Take 200ml of sample in 26 bottles.















Add standard Chlorine solution to the bottles through pipette in 0.5ml increment.















Squeeze the pipette bulb and dip the pipette into the Chlorine solution
Press the up arrow on the bulb to take the liquid up into the pipette
Press the down arrow on the bulb to release chlorine solution
0ml
Mix the sample by inverting the bottle and keep all the bottles in dark for 20 minutes.






Take out the bottles after 20 minutes
Pour 200ml of sample from each bottles into conical flask.







Add 5ml of concentrated Sulphuric Acid (H2SO4) to the flask.








Squeeze the pipette bulb and dip the pipette into H2SO4 solution
Press the up arrow on the bulb to take the liquid up into the pipette
Add approximately 1g Potassium Iodide (KI) crystals and stirr the flask properly.




Add 1ml of Starch indicator to conical flask and observe the change in colour of the solution from colourless to pale blue.





Take 0.0282N Iodine solution in the burette.










Titrate the 0.5ml solution against Iodine solution.










Open the knob to start the liquid running into the conical flask
Close the knob when the colour of solution in conical flask changes to pale pink
Observe the colour change from pale blue to pale pink
Add 1ml Starch into conical flask
Initial burette reading = 0 ml
Observations:
| Chlorine dosage (mg/l) | Burette Reading (ml) | Volume of Iodine solution ( Final Value - Initial Value ) (ml) | |
| Initial | Final | ||
✔
✘

Titrate the solution with 1ml Chlorine solution against 0.0282N Iodine solution.











Open the knob to start the liquid running into the conical flask
Close the knob when the colour of solution in conical flask changes to pale pink
Observe the colour change from pale blue to light pink
Add 1ml Starch into conical flask
Initial burette reading = 0ml
2. Add 5ml of H2SO4 to it
3. Add 1g Potassium Iodide (KI) crystals and stirr the flask properly4. Add 1ml of Starch indicator to flask5. Titrate it with 0.0282N Iodine solution
Observations:
| Chlorine dosage (mg/l) | Burette Reading (ml) | Volume of Iodine solution ( Final Value - Initial Value ) (ml) | |
| Initial | Final | ||
✔
✘

Titrate the 1.5ml solution against 0.0282N Iodine solution.











Open the knob to start the liquid running into the conical flask
Close the knob when the colour of solution in conical flask changes to pale pink
Observe the colour change from pale blue to pale pink
Add 1ml Starch into conical flask
Initial burette reading = 0ml
2. Add 5ml of H2SO4 to it
3. Add 1g Potassium Iodide (KI) crystals and stirr the flask properly4. Add 1ml of Starch indicator to flask5. Titrate it with 0.0282N Iodine solution
Observations:
| Chlorine dosage (mg/l) | Burette Reading (ml) | Volume of Iodine solution ( Final Value - Initial Value ) (ml) | |
| Initial | Final | ||
✔
✘

Observations:
Characteristic Curves
Chlorine dosage(mg/l) v/s Residual chlorine(mg/l)
Break point chlorine of water sample = mg/l
Chlorine demand of water sample = mg/l
✔
✔
✘
✘
Inference:
The acceptable range of residual chlorine in water is
less than 0.2mg/l between 0.2mg/l and 0.5mg/l 0.5mg/l - 1mg/l greater than 1mg/lTitration =